Waste pork bones turned into an adsorbent for toxic metals
—Development of low-cost, high-performance metal adsorbent technology using food waste—
A growing number of preventive measures than before are being implemented around mining and industrial areas to deal with environmental pollution caused by hazardous metals. Since the nuclear accident at Fukushima Daiichi Nuclear Power Plant in the wake of the Tōhoku earthquake and tsunami, there has been renewed awareness regarding the importance of technologies for preventing leaks and widespread dispersal. To prevent hazardous metals from spreading over an extensive environmental area, there is a need for developing a method capable of producing large quantities of low-cost and high-performance adsorbents.
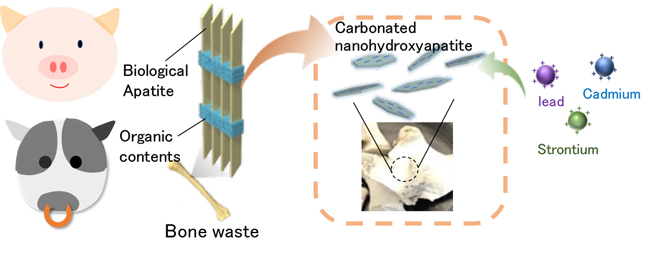
Beef and pork bones that are discarded in large quantities as food waste are known to have metal adsorption characteristics. However, the metal adsorptivity of bones is not higher than other conventional adsorbents such as clays and zeolites. It was needed to improve of metal adsorptivity of bones for effective use of bone waste.
Immersing discarded pork bones in an aqueous sodium bicarbonate solution produces high carbonated apatite. This carbonated apatite adsorbs strontium ions 250-times more than unprocessed bones and 20-times more than natural zeolite adsorbents that are used as strontium adsorbents, and also indicated a high adsorption performance for cadmium and lead.
This extremely easy method was used to successfully develop an environmental decontamination material made from discarded bones found in common food waste. This achievement is expected to be used in applications like purifying polluted water, environmental cleanup efforts and also recovery processes for rare metals.