Supplementary document
1. Background
JAERI (Japan Atomic Energy Research Institute), one of the former
institutes of JAEA, has selected novel technologies with higher
potentials for industrial use, and exhibited them through such
as technology fairs, in order to actively transfer the R&D
results to industrial entities.
In this development, we have actively engaged in the whole process of:
- Selection of promising technologies for practical applications.
- Introducing them at technology-transfer fairs and at catalogues.
- Detailed technical consultation for visited companies.
- Contract and execution of patent enforcement consents and technical
guidances.
- Set up a special team for executing a definite support to manufacturers
that will commercialize.
- Further joint development meet for demand of product specifications
from customers.
- Keep confidential if necessary.
In terms of the effective use of intellectual property rights,
there were formerly a few examples of the practical use of patent
enforcement consents to companies. However, due to insufficient
participation of the research organization side for the necessary
follow-up development, an essential effort for the utilization/commercialization
was implemented mainly by the company side, so that contribution
by the organization side was reduced or underestimated. Based on
these reconsideration, we actively and systematically work as a
development project of the industry-organization co-operation, particularly
for the technology in which the utilization/commercialization is
highly anticipated.
2. Social contribution by commercialization
The main characteristic of this technology is that we can measure
a weight of the impurity gas released from measuring objects such
as aluminum casting parts, with a small sample, a short time and
a high accuracy, by using a small movable device.
This technology opens the new application of quality control not
only for the meltdown aluminum, also for other many materials.
It is also expected to deeply contribute for securing security
and safety of cast products widely used in our life.
3. Technical contents
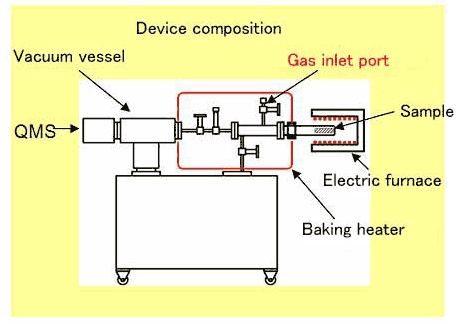
The new “Gravi-Mass” we have developed, consists of
an electric furnace that heats a sample metal such as aluminum,
a vacuum chamber and a gas analysis sensor (Quadrupole Mass Spectrometer:
QMS). This system can analyze many sort of gases released from
a metal sample at the same time. A direct analysis of the sample
gas to be measured is also possible by introducing the gas to
an inlet port.
In a conventional gas analysis method, the gas quantity was expressed
by density which is the volume value (cm3/g), however, a precise
monitoring of a circumference such as temperature and pressure
was indispensable to calculate the gas quantity, because the volume
value was, in principle, influenced severely by the temperature
and the pressure (a gas volumetry method). Therefore, this method
has an intrinsic disadvantage of deteriorating the accuracy, when
the sample gas temperature and pressure fluctuate during analysis.
In addition, when we compare the results of measurement values
among analyzing organizations, the measurement conditions should
be converted by the same standard.
The new Gravi-Mass method we have developed, measures the gas
quantity by its weight. This absolute measurement is no need to
consider the fluctuation of the gas temperature and pressure.
Therefore, it has been possible to secure the traceability (history
management) which can reproduce the measured value without being
influenced by the measurement conditions (refer to the table below).

| Influencing factor |
Gas volmetry method |
Gravi-Mass method |
| Circumference (temperature, pressure) |
The fluctuations of values should be monitored during a measurement time, in every measurement day and measurement place, and the values measured should be converted by the same standard. |
No need to consider. |
| Sample temperature |
A temperature of the sample gas varies with temperature fluctuations of the device, which causes an error of temperature. |
Not influenced. |
| Sample pressure |
In case a number of unknown gases are included, it causes a pressure error because it is actually impossible to identify the partial pressures of all gases. |
Not influenced. |
| |
At present, it applies only to hydrogen. |
|
 Back to the article
Back to the article






